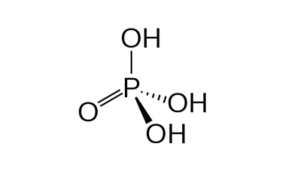
Phosphoric Acid (Technical)
CAS : 7664-38-2
Phosphoric Acid (Technical)
DESCRIPTION
Phosphoric Acid is a colorless, odorless phosphorus-containing inorganic acid. It has a role as a solvent, a human metabolite, an algal metabolite and a fertilizer. It is miscible with water, ethanol and mixture of ether: alcohol (3:1).
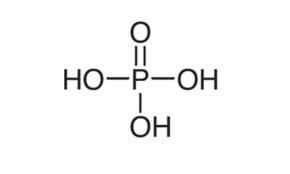
PROPERTIES
| Appearance | Transparent Colorless Fluid |
| Actual Active Ingredient (H3PO4) | 85.10 % |
| Chloride ( as Cal) | 0.0003 % |
| Sulphate (as SO4) | 0.003 % |
| Heavy Metal (as Pub) | N.D |
| Arsenic ( as As) | N.D |
| Iron (as Fe) | 0.0003 % |
| Acetic Acid ( CH3COOH) | 1.94 % |
APPLICATION
· Phosphoric acid is used mainly in fertilizers.
· It is used as an external standard for phosphorus-31 Nuclear magnetic resonance (NMR)
· It is used as an electrolyte in phosphoric acid fuel cells and as a catalyst in the hydration of alkenes to produce alcohols, predominantly ethanol.
· It finds application as a buffer agent in biology and chemistry; as a solution for anodizing and as a cleaner by construction trades to remove mineral deposits, cementations smears and hard water stains.
· It is also used as a pH adjuster in cosmetics and skin-care products and a dispersing agent in detergents and leather treatment.
· It plays an important role in the preparation of albumin derivatives.
· It is an important starting material used in the preparation of disodium hydrogen phosphate, disodium phosphate, calcium hydrogen phosphate and monohydrate phosphate.
· It is widely used to remove rust in steel tools or surfaces by converting the rusted ion in to water soluble phosphate compound.
PACKAGING OPTIONS
Drums
Enquiry Now