Dimethyl Sulfoxide
Dimethyl Sulfoxide
DESCRIPTION
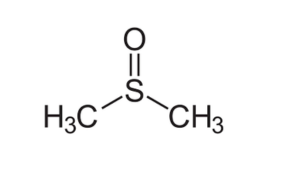
Dimethyl sulfoxide is a 2-carbon sulfoxide in which the sulfur atom has two methyl substituents. Dimethyl sulfoxide appears as a clear liquid, essentially odorless. It is a highly polar organic liquid that is used widely as a chemical solvent and a free radical scavenger.
PROPERTIES
| Purity (DMSO) % | 99.93 |
| Crystallization Point °C | 18.34 |
| Acid Value MGKOH/G | 0.02 |
| Moisture % | 0.02 |
| Transmittance (400MM)% | 99.00 |
| Refractive Index (20°C) | 1.4786 |
APPLICATION
· DMSO is frequently used as a solvent for chemical reactions involving salts, most notably Finkelstein reactions and other nucleophile substitutions.
· It is also extensively used as an extract ant in biochemistry and cell biology.
· DMSO is used in polymerase chain reaction (PCR) to inhibit secondary structures in the DNA template or the DNA primers.
· DMSO may also be used as a cry protectant, added to cell media to reduce ice formation and thereby prevent cell death during the freezing process.
· medicine, DMSO is predominantly used as a topical analgesic, a vehicle for topical application of pharmaceuticals, as an anti-inflammatory, and an antioxidant.[19] Because DMSO increases the rate of absorption of some compounds through biological tissues, including skin, it is used in some transdermal drug delivery systems.
PACKAGING OPTIONS
Drums
Enquiry Now