Ethyl Acetate
CAS : 141-78-6
Ethyl Acetate
DESCRIPTION
Ethyl acetate is the acetate ester formed between acetic acid and ethanol with a molecular formula of C4H8O2. It is a colorless liquid with a fruity characteristic odor that is commonly recognized in glues and nail polish remover. This product is highly flammable and it is miscible with all common organic solvents (alcohols, ketones, glycols, esters) but only slightly miscibility in water.
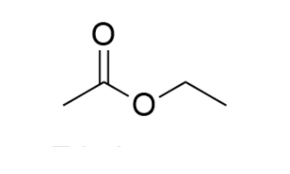
PROPERTIES
| Water | 120 mg/kg |
| Acidity (as Acetic Acid) | 21 mg/kg |
| Platinum Cobalt Color | 5 |
| Relative Density at 20/20°C | 0.9021 |
| Distillation Range of Volatile Organic Liquid | - |
| Initial Boiling Point | 76.9 °C |
| Dry Point | 77.5 °C |
| Distillation Range | 0.6 °C |
| Non-Volatile Matter | < 1 mg/100mL |
| Residual Odor | Non-Residual |
| Alcohol Content and Purity of Acetate Esters by GC | - |
| Ethanol | 100 mg/kg |
| Ethyl Acetate | 99.94 %(m/m) |
APPLICATION
· In the field of entomology, ethyl acetate is an effective asphyxiant for use in insect collecting and study.
· Ethyl acetate is most commonly used as a solvent (due to its dilution properties).
· As a high purity solvent, it is used in cleaning electric circuit boards and as a nail polish remover.
· At a lower purity, it can be used as in printing, pharmaceuticals, perfumes, food, decaffeination of tea/coffee and a carrier solvent for herbicides.
· Ethyl acetate is also used in coating formulations for wood furniture, agricultural, construction equipment, mining equipment and marine uses.
· It is also naturally produced in wine during the fermentation process.
· The main user end markets of these products are the electronics, cosmetic, printing, food and coatings industries.
PACKAGING OPTIONS
Drums
Enquiry Now