N-Butyl Bromide
CAS : 109-65-9
N-Butyl Bromide
DESCRIPTION
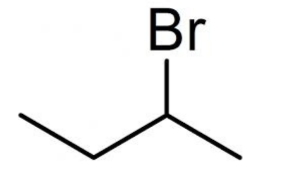
N-Butyl Bromide is the organobromine compound with the formula CH3(CH2)3Br. It is a colorless liquid, although impure samples appear yellowish. It is insoluble in water, but soluble in organic solvents. It is a primarily used as a source of the butyl group in organic synthesis. It is one of several isomers of butyl bromide.
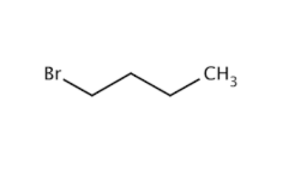
PROPERTIES
| Appearance | Colourless liquid |
| Density | 1.2676 g mL−1 |
| Melting point | −112.5 °C; −170.4 °F; 160.7 K |
| Boiling point | 101.4 to 102.9 °C; 214.4 to 217.1 °F; 374.5 to 376.0 K |
APPLICATION
It is commonly used as an alkylating agent. When combined with magnesium metal in dry ether, it gives the corresponding Grignard reagent. Such reagents are used to attach butyl groups to various substrates.
PACKAGING OPTIONS
Drums
Enquiry Now