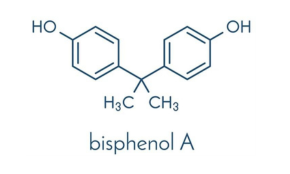
Bisphenol A
CAS : 80-05-7
Bisphenol A
DESCRIPTION
Bisphenol a (BPA) is an organic synthetic compound with the chemical formula (CH3)2C(C6H4OH) belonging to the group of diphenylmethane derivatives and bisphenols, with two hydroxyphenyl groups. It is colorless solid that is soluble in organic solvents, but poorly soluble in water. It is a dysfunctional building block of several important plastics and plastic additives. With an annual production of 2–3 million metric tons, it is an important monomer in the production of polycarbonate.
PROPERTIES
| Grade | BPA |
| Purity (dry basis) (wt. %) | 99.94 |
| Phenol (wt.-ppm) | 6 |
| Water (wt. %) | 0.047 |
| Freezing Point (°C) | 156.6 |
| Methanol Color (APHA) | 1.9 |
| Iron (wt.-ppm) | 0.01 |
| Ash (wt.-ppm) | < 5 |
APPLICATION
· The largest use for bisphenol A is polycarbonate followed by epoxy resins. Other uses are flame retardants, unsaturated polyester resins and polyacrylate, polyetherimide, and polysulfone resins.
· Adhesives and sealant chemicals
· Intermediates
· Laboratory chemicals
· Paint additives and coating additives not described by other categories
· Process regulators
· Propellants and blowing agents
PACKAGING OPTIONS
Drums
Enquiry Now